From Pure Blockchain to Pragmatic Compliance-First Solution
TraceVida was born as a response to the Medifarma crisis in March 2025, where contaminated saline solution with sodium concentrations up to 600% higher than permitted caused the death of seven people in Peru, including a one-year-old baby (France 24, 2025), exposing critical systemic failures in Peruvian pharmaceutical traceability (Pinedo, 2025). The initial vision proposed using blockchain technology to create an immutable end-to-end traceability system from manufacturer to patient.
The initial proposal promised registration of each pharmaceutical batch on public blockchain, QR codes with transaction IDs for instant verification, and a competitive cost model compared to existing solutions based on Ethereum, where implementations like PharmaChain 3.0 report costs of ~$2.2 USD for factory contract deployment, ~$1.6 USD for lot contract generation, and an average of $0.86 per transaction (Bapatla et al., 2022, p. 630).
Peru’s Law No. 29733 on Personal Data Protection allows cross-border data transfer when the recipient country has an adequate level of protection or when adequate guarantees are provided (Ministry of Justice and Human Rights, 2024, art. 18). However, the Latin American region is rapidly converging toward the European GDPR model, making pure blockchain solutions a commercially suicidal strategy.
The Regulatory Convergence Trap:
Brazil, the largest Latin American market, is in the final phase of mutual recognition with the European Union. The Autoridade Nacional de Proteção de Dados (ANPD) is working on issuing an adequacy decision recognizing the equivalence between LGPD and GDPR (Autoridade Nacional de Proteção de Dados, 2025), while the European Commission has already published the preliminary version recognizing that Brazil offers a level of protection equivalent to the European one. This convergence de facto establishes GDPR as the regional standard.
The Schrems II precedent from the Court of Justice of the European Union invalidated international transfer agreements due to insufficient guarantees, with significant effects for health research and personal data transfers (Hallinan et al., 2021). A startup that adopts pure blockchain architecture today closes its expansion path to Brazil and, eventually, to the rest of LATAM that will follow the GDPR-aligned model.
Insurmountable Practical Consequences:
• Hashes of patient information on public blockchain constitute personal data under GDPR (European Data Protection Board, 2025, p. 12) • Impossibility of complying with the ‘right to be forgotten’ due to blockchain immutability, as information remains permanently recorded in geographically distributed blocks (Buleje, 2022) • Cost of re-architecting post-traction >> cost of designing correctly from the start • Regional expansion blocked by inevitable adoption of GDPR-aligned standards
Analysis of the Latin American pharmaceutical market revealed that the main obstacle to traceability system adoption is not lack of technology, but cost structure and integration with existing operational processes (Silva & Mattos, 2019).
Priority Operational Problems:
Independent pharmacies in emerging markets face critical operational challenges that supersede traceability concerns in priority. Implementation of traceability systems requires doubling human resources, acquiring additional technology (readers, hardware), and integrating software that is frequently not ready, significantly increasing bureaucratic burden without necessarily improving patient dispensation (Confederación Farmacéutica Argentina [COFA], 2016; Administración Nacional de Medicamentos, Alimentos y Tecnología Médica [ANMAT], 2016). In contexts where two-thirds of medication expenditure comes directly from household pockets (Tobar & Martich, 2014), pharmacies naturally prioritize operational efficiency and access over complex traceability systems.
• Significant cash flow loss due to delays in distributor payments and informal credit systems. In Argentina, collection delays have exacerbated the delicate situation of small and medium pharmacies, where loss of operability and stock leads clients to resort to larger pharmacies with greater economic capacity (Conti, 2014). Similarly in Costa Rica, verticalization of the distribution chain has resulted in independent pharmacies having less muscle to obtain better prices, with distributors granting preferential conditions to their own chains (Molina, 2023)
• Inventory waste due to expiration of medications without adequate rotation. In developing countries, inadequate monitoring of expiration times and inventory management problems result in waste rates of up to 3.68%, with 92% of waste attributable to expiration (Ayenew et al., 2021). In Latin America, hospital studies in Costa Rica confirm that 99.6% of discarded medications correspond to expired products (Zavaleta-Monestel et al., 2024)
• Internal theft and fraud. In the United States, employee theft represents 46% of pharmacy thefts (Drug Enforcement Administration, 2013), with estimated pharmaceutical fraud losses of $3.5 billion annually (Timofeyev et al., 2022). In Latin America, internal theft in the retail sector represents 42.6% of total losses, with employees stealing on average eight times more than customers (Bamfield, 2011); these figures would presumably be higher in pharmacies given the high resale value, portability, and ease of monetization of medications.
• Competition with counterfeit medications with an illegal market estimated at $200 million annually, representing approximately 13% of the Peruvian pharmaceutical market valued at $1,500 million (Videnza Consultores, 2019).
• Manual credit management with records in physical notebooks without digitization; informal credit (“fiado”) represents the second most used form of informal credit in low-income strata in Colombia (Martinez Benavides, 2021).
Independent pharmacies in emerging markets prioritize solutions to concrete operational problems—internal theft, illegal competition, informal credit—over technological features that Silicon Valley would consider priorities.
This operational prioritization has direct implications for product-market fit: pharmacies don’t buy blockchain technology—they buy reduction of theft losses, protection against illegal competition, and tools to manage informal credit. The value proposition must be articulated in terms of these concrete problems, not immutability or decentralization.
Strategic Decision: Pivot from ‘blockchain traceability platform’ toward ‘WhatsApp-first logistics aggregator’ with optional blockchain verification capabilities implemented only after achieving significant network effect.
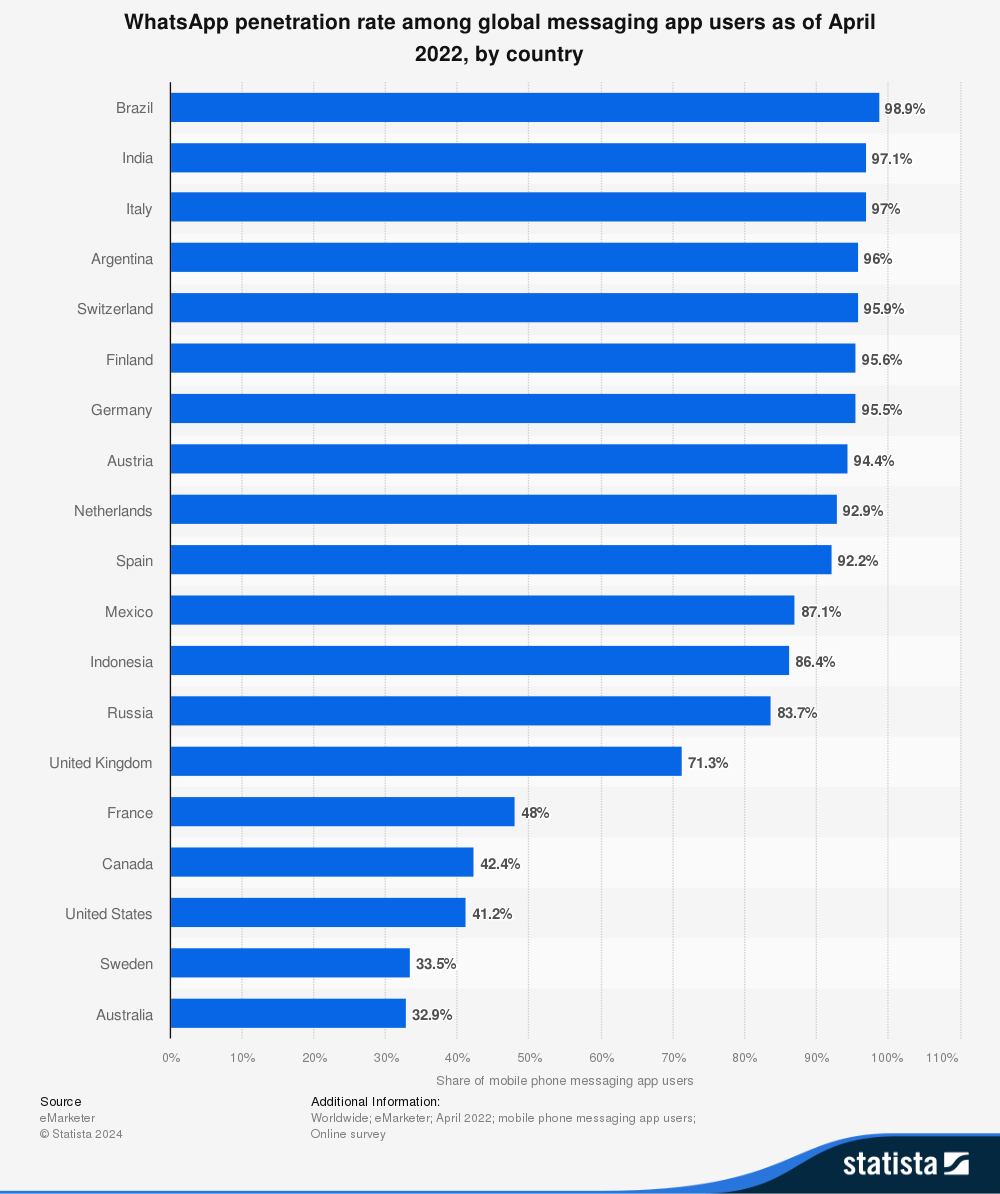
The WhatsApp-first strategy responds to the reality of the Latin American market, where Brazil leads globally with 98.9% penetration among messaging app users, followed by Argentina with 96% and Mexico with 87.1% (eMarketer, 2022). This existing social infrastructure eliminates technology adoption barriers that dedicated native platforms would face, allowing immediate integration with daily workflows of independent pharmacies without requiring training or installation of new applications.

Figure 1. WhatsApp penetration rate among messaging app users by country. Note. From "WhatsApp penetration rate among global messaging app users as of April 2022, by country" [Graph], by eMarketer, 2022, Statista (https://www.statista.com/statistics/1311229/). Copyright 2024 by Statista.
This strategy prioritizes solving immediate operational problems that generate revenue (order management, inventory, delivery) and relegates blockchain to a later phase when there is critical mass of users.
Digital infrastructure in Peru presents significant barriers for deployment of systems requiring constant connectivity. A study of real deployment in 6 communities in the Peruvian Amazon documents that the connectivity gap in rural areas is caused by urban-centric business models that are not cost-effective in regions characterized by isolation, low population density, and resource scarcity (Prieto-Egido et al., 2020).
Pharmaceutical supply chains extending to remote areas face connectivity challenges exacerbated by mountainous geography and tropical jungle. Unlike messaging applications that function with intermittent connectivity through store-and-forward architectures, the blockchain consensus mechanism requires reliable and constant connectivity for all participating nodes, creating critical operational vulnerability in contexts where communications infrastructure is nonexistent or intermittent.
Layer 2 architecture (audit logs with hash chains, described in detail in section 3.4) provides sufficient immutability and traceability for regulatory compliance without depending on public blockchain. The main difference between Layer 2 and Layer 3 (optional blockchain anchor) is independent external verifiability, not immutability itself.
Layer 2 provides cryptographic proof of non-modification through hash chains, complete audit trail from manufacturer to patient, compliance with DIGEMID/APEMED requirements, and verification by regulatory authorities. Layer 3 adds verification by any third party without system access, resistance to operator coercion, and marketing differentiation (“blockchain-anchored”).
Postponing blockchain does not sacrifice traceability—it only postpones public verifiability until the network effect justifies the additional investment.
Academic debate on private blockchain versus traditional databases has established that both technologies functionally converge when control is centralized in one entity. As Narayanan (2015) states, private blockchains lose the security guarantees that come from decentralization and proof-of-work, effectively functioning as shared databases with cryptographic features.
Security guarantees of distributed ledgers and private blockchains depend on the honesty of entities validating transactions, without mathematical guarantees of irreversibility as in public blockchains.
The decision between blockchain and traditional database should be based on analysis of trust boundaries between system participants. Wüst and Gervais (2018) establish that blockchain only makes sense when multiple entities without mutual trust need to interact and modify the state of a system. When there is only one writer, blockchain provides no additional guarantees and a traditional database is superior in performance.
Analysis for TraceVida:
• No trust boundary between pharmacies and aggregator platform (established contractual relationship) • No trust boundary between customer and pharmacy (regulatory trust via DIGEMID license) • There is a potential trust boundary between government/regulator and pharmaceutical industry
The latter can be resolved with local immutable audit logs without requiring external blockchain.
In centralized systems, performance in latency and throughput is significantly superior to blockchain systems due to the additional complexity of the consensus mechanism (Wüst & Gervais, 2018). Bitcoin currently sustains approximately seven transactions per second, while centralized systems like Visa handle peaks of more than fifty thousand transactions. There is an inherent trade-off between decentralization—how well a system scales to multiple writers without mutual trust—and throughput.
Overhead Factors:
• Digital signature verification for each transaction • Consensus among multiple nodes • Processing redundancy (each node processes each transaction)
The proposed architecture operates in three layers:
• Traditional database - high-frequency operations and GDPR compliance • Audit logs with hash chains - traceability without blockchain • Blockchain anchor (optional) - external verification post-network effect
This separation allows optimization for different objectives simultaneously: legal compliance (Layer 1), internal traceability (Layer 2), and decentralized verification when the market justifies it (Layer 3), as illustrated in Figure 2.
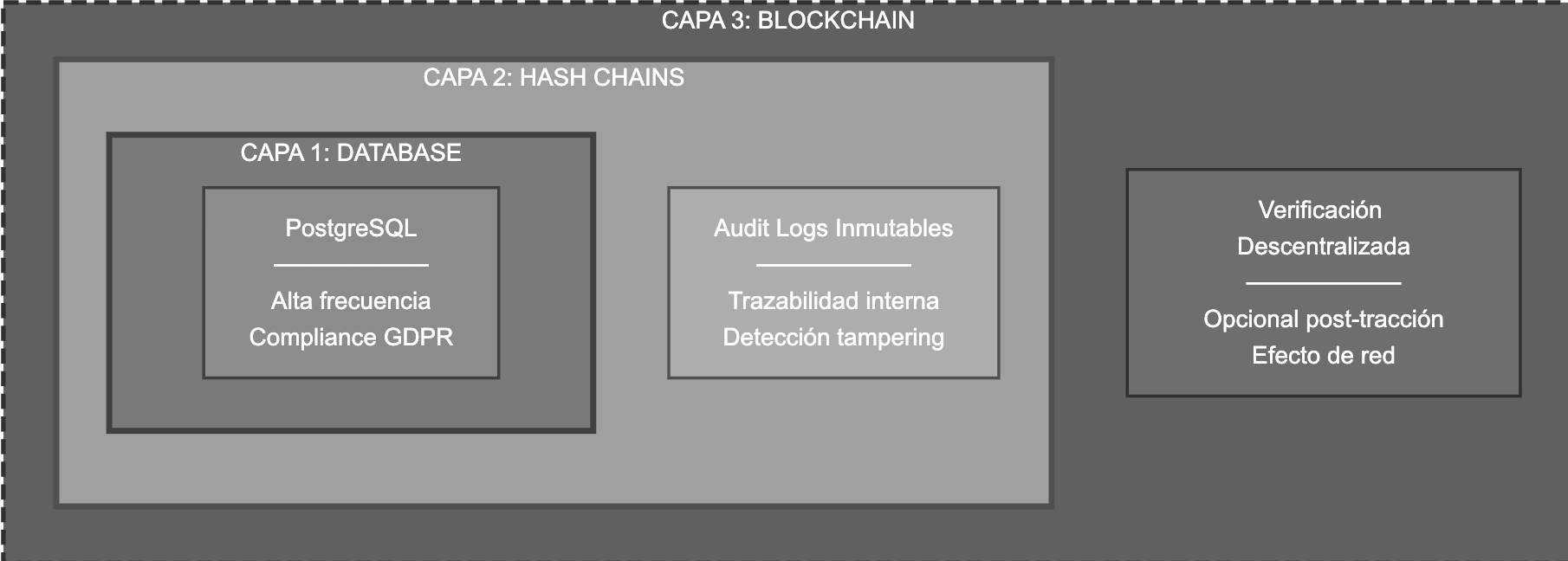
Figure 2. Three-layer architecture of TraceVida. Own elaboration.
The proposed architecture provides legal compliance by maintaining personal data off-chain in appropriate jurisdiction, allowing compliance with right to be forgotten without compromising hash chain immutability. Without distributed consensus overhead, the system achieves superior performance without transaction fees in high-frequency operations. Append-only logs with hash chains provide verifiable auditability, while optional blockchain anchor allows independent external verification when network effect justifies it.
The TraceVida case illustrates a fundamental lesson about adoption of emerging technologies in regulated markets: technology must serve the business and regulatory framework, not vice versa. The solution is not to abandon the benefits of immutability and traceability that blockchain promises, but to implement them pragmatically in a way that respects legal, operational, and economic constraints of the real context.
In emerging markets with strict regulations, pragmatism is not compromise—it is survival.
Administración Nacional de Medicamentos, Alimentos y Tecnología Médica. (2016, 23 de septiembre). Disposición 10564/2016 [Sistema Nacional de Trazabilidad de Medicamentos]. Boletín Oficial de la República Argentina. https://www.boletinoficial.gob.ar/detalleAviso/primera/151275/20160923
Autoridade Nacional de Proteção de Dados. (2025, 5 de septiembre). Unión Europea divulga versión preliminar de decisión de adecuación. https://www.gov.br/anpd/pt-br/assuntos/noticias/union-europea-divulga-version-preliminar-de-decision-de-adecuacion
Ayenew, W., Asmamaw, G., Kebede, N., & Mullatu, G. (2021). Pharmaceuticals wastage and pharmaceuticals waste management in public health facilities of Dessie town, North East Ethiopia. PLOS ONE, 16(11), Article e0259160. https://doi.org/10.1371/journal.pone.0259160
Bamfield, J. (2011, October 19). Global Retail Theft Barometer study finds shrink at highest levels since 2007. Centre for Retail Research. https://www.retailitinsights.com/doc/global-retail-theft-barometer-study-finds-0003
Bapatla, A. K., Mohanty, S. P., Kougianos, E., & Puthal, D. (2022). PharmaChain 3.0: Blockchain integrated efficient QR code mechanism for pharmaceutical supply chain. 2022 OITS International Conference on Information Technology (OCIT), 625-630. https://doi.org/10.1109/OCIT56763.2022.00121
Buleje, C. (2022). La Blockchain y los retos en la aplicación de las leyes de protección de datos personales. Prometheo CDA. https://prometheo.pe/la-blockchain-y-los-retos-en-la-aplicacion-de-las-leyes-de-proteccion-de-datos-personales/
Confederación Farmacéutica Argentina. (2016, 30 de septiembre). La nueva disposición sobre Trazabilidad responde al reclamo de los farmacéuticos [Comunicado de prensa]. https://portal.cofa.org.ar/2016/09/30/la-nueva-disposicion-sobre-trazabilidad-responde-al-reclamo-de-los-farmaceuticos/
Conti, O. O. (2014, 4 de agosto). Diagnostico organizacional: Caso farmacias pequeñas en Argentina [Entrada de blog]. Prácticas Farmacéuticas. https://blogs.ead.unlp.edu.ar/practicafarmaceutica/2014/08/04/diagnostico-organizacional-caso-farmacias-pequenas-en-argentina/
Drug Enforcement Administration. (2013, junio). Drug theft prevention [Diapositivas de PowerPoint]. Office of Diversion Control. Archivado en DocumentCloud por N. Schalit & J. Christie. https://www.documentcloud.org/documents/782120-dea-publication-on-pharmacy-thefts
eMarketer. (2022). WhatsApp penetration rate among global messaging app users as of April 2022, by country [Gráfico]. Statista. https://www.statista.com/statistics/1311229/whatsapp-usage-messaging-app-users-by-country/
European Data Protection Board. (2025). Guidelines 02/2025 on processing of personal data through blockchain technologies (Version 1.1). https://www.edpb.europa.eu/system/files/2025-04/edpb_guidelines_202502_blockchain_en.pdf
France 24. (2025, 25 de abril). Un suero mal fabricado ya mató a siete personas en Perú y aún no hay responsables. https://www.france24.com/es/américa-latina/20250425-un-suero-mal-fabricado-ya-mató-a-siete-personas-en-perú-y-aún-no-hay-responsables
Hallinan, D., Bernier, A., Cambon-Thomsen, A., Crawley, F. P., Dimitrova, D., Medeiros, C. B., Nilsonne, G., Parker, S., Pickering, B., & Rennes, S. (2021). International transfers of personal data for health research following Schrems II: A problem in need of a solution. European Journal of Human Genetics, 29(10), 1502-1509. https://doi.org/10.1038/s41431-021-00893-y
Martinez Benavides, A. (2021). Circuitos crediticios: el fiado en Cali, Colombia. Estudios Sociológicos, 39(116), 467-494. https://doi.org/10.24201/es.2021v39n116.1946
Ministerio de Justicia y Derechos Humanos. (2024, 30 de noviembre). Decreto Supremo N.° 016-2024-JUS, Reglamento de la Ley N.° 29733, Ley de Protección de Datos Personales. Diario Oficial El Peruano. https://img.lpderecho.pe/wp-content/uploads/2024/11/Decreto-Supremo-016-2024-JUS-LPDerecho.pdf
Molina, L. (2023, 15 de noviembre). Farmacias independientes pierden terreno frente al voraz crecimiento de las cadenas. Bitácora Farmacéutica. https://bitacorafarmaceutica.com/noticias/farmacias-independientes-pierden-terreno-frente-al-voraz-crecimiento-de-las-cadenas/
Narayanan, A. (2015, September 18). “Private blockchain” is just a confusing name for a shared database. CITP Blog. https://blog.citp.princeton.edu/2015/09/18/private-blockchain-is-just-a-confusing-name-for-a-shared-database/
Pinedo, X. (2025, 13 de abril). Caso Medifarma: el suero defectuoso que expuso las grietas del sistema sanitario. Ojo Público. https://ojo-publico.com/derechos-humanos/salud/medifarma-el-caso-que-expuso-las-grietas-del-sistema-sanitario
Prieto-Egido, I., Valladares, J. A., Muñoz, O., Bernuy, C. C., Simo-Reigadas, J., Quispetupa, D. A., Fernández, A. B., & Martinez-Fernandez, A. (2020). Small rural operators techno-economic analysis to bring mobile services to isolated communities: The case of Peru Amazon rainforest. Telecommunications Policy, 44(10), Article 102039. https://doi.org/10.1016/j.telpol.2020.102039
Silva, R. B. D., & Mattos, C. A. (2019). Critical success factors of a drug traceability system for creating value in a pharmaceutical supply chain (PSC). International Journal of Environmental Research and Public Health, 16(11), 1972. https://doi.org/10.3390/ijerph16111972
Timofeyev, Y., Hayes, S. A., & Jakovljevic, M. B. (2022). Predictors of loss due to pharmaceutical fraud: Evidence from the U.S. Cost Effectiveness and Resource Allocation, 20, Article 6. https://doi.org/10.1186/s12962-022-00337-4
Tobar, F., & Martich, E. (2014). Economic tools for ensuring access to medicines in Latin American countries. Pharmaceuticals Policy and Law, 16(3-4), 207-224. https://doi.org/10.3233/PPL-140385
Videnza Consultores. (2019). Medicamentos ilegales en el Perú: Diagnóstico de la situación y recomendaciones de política [Informe final]. Asociación Nacional de Cadena de Boticas (ANACAB).
Wüst, K., & Gervais, A. (2018). Do you need a blockchain? 2018 Crypto Valley Conference on Blockchain Technology (CVCBT), 45-54. IEEE. https://doi.org/10.1109/CVCBT.2018.00011
Zavaleta-Monestel, E., Cartín-Ramírez, M. A., Rojas-Chinchilla, C., Arguedas-Chacón, S., & Villalobos-Madriz, J. A. (2024). Management of medications disposal and the economic burden in a sustainable Latin American hospital. Sustainable Chemistry and Pharmacy, 39, Article 101540. https://doi.org/10.1016/j.scp.2024.101540